GDF-8 Injection gold bond
GDF-8 Injection gold bond Specification
- Dosage Form
- Injection
- Pacakaging (Quantity Per Box)
- 1 vial per box
- Indication
- Primary indication for advanced muscle wasting therapies and research
- Origin of Medicine
- Biotechnological synthesis
- Salt Composition
- Recombinant GDF-8 analogue
- Drug Type
- Biological injectable
- Ingredients
- Recombinant Myostatin (GDF-8) analogues, sterile water for injection, stabilizing agents
- Physical Form
- Liquid
- Function
- Promotes muscle hypertrophy by inhibiting myostatin activity
- Recommended For
- Muscle growth, performance enhancement, research purposes
- Dosage
- 1 mg per vial (as per vial marking)
- Dosage Guidelines
- As directed by healthcare professional or researcher
- Suitable For
- Adults
- Quantity
- 1 vial per box
- Storage Instructions
- Store in a cool, dry place at 2-8C; protect from light
- Caution
- For research use or prescribed professional administration only; not for self-administration
- Molecular Weight
- Approx. 26 kDa (GDF-8 protein)
- Route of Administration
- Intramuscular or subcutaneous injection
- Registration Status
- Not for retail sale, laboratory/clinical research use only
- Appearance
- Clear to light yellow liquid
- Shelf Life
- 24 months from manufacturing date (when stored as instructed)
- Sterility
- Sterile, single-use vial
GDF-8 Injection gold bond Trade Information
- Supply Ability
- 100 Per Day
- Delivery Time
- 7 Days
- Sample Available
- No
- Sample Policy
- Contact us for information regarding our sample policy
About GDF-8 Injection gold bond
Precise Muscle Growth Enhancement
GDF-8 Injection Gold Bond works by inhibiting myostatin activity, which in turn promotes muscle hypertrophy. This action makes it valuable for research into muscle wasting therapies and performance enhancement studies. The standardized 1 mg dosage per vial allows for consistent experimental results, provided the administration is conducted by qualified professionals.
Safe and Controlled Application
Each sterile vial is intended for single use and should be administered either intramuscularly or subcutaneously, as directed by a healthcare professional or qualified researcher. Laboratory or clinical settings are recommended for its use, ensuring adherence to safety protocols and regulatory guidelines. The formulation remains stable for up to 24 months when stored correctly.
FAQ's of GDF-8 Injection gold bond:
Q: How should GDF-8 Injection Gold Bond be stored for maximum shelf life?
A: Store the injection in a cool, dry place at temperatures between 2-8C, ensuring it is protected from light. Proper storage conditions help maintain sterility and effectiveness for up to 24 months from the manufacturing date.Q: What is the recommended route of administration for this injection?
A: GDF-8 Injection Gold Bond is designed for intramuscular or subcutaneous injection, and should be administered by a qualified healthcare professional or researcher in a controlled setting.Q: Can individuals self-administer this product for muscle growth?
A: No, self-administration is not recommended. This product is intended only for research use or prescribed professional administration, and should not be used without appropriate supervision.Q: Who is suitable to receive GDF-8 Injection Gold Bond?
A: The product is suitable for adults participating in approved research studies or advanced therapeutic protocols for muscle wasting. It is not available for retail or general consumer use.Q: What are the key benefits of using GDF-8 Injection Gold Bond in research applications?
A: The primary benefit is its ability to promote muscle hypertrophy via myostatin inhibition, making it valuable for studying muscle growth, performance enhancement, and muscle wasting conditions.Q: Where should the injection be performed to ensure safety and compliance?
A: Administration should take place in laboratory or clinical environments under professional supervision, strictly adhering to institutional safety guidelines and regulatory requirements.Q: What process should be followed for dosage and administration?
A: Use the 1 mg (per vial) dosage as marked, following specific instructions provided by a healthcare professional or designated researcher. Exact dosage may vary depending on the research protocol or treatment plan.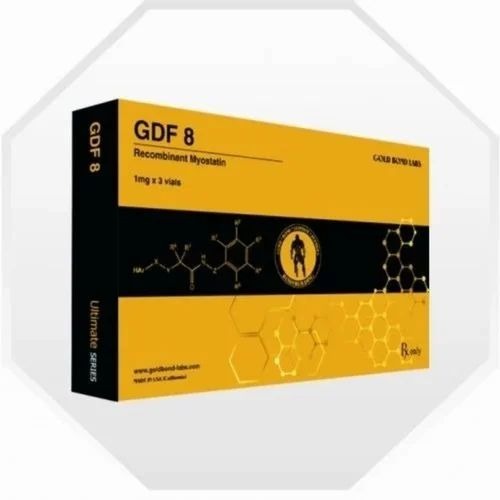

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharmaceutical Injection Category
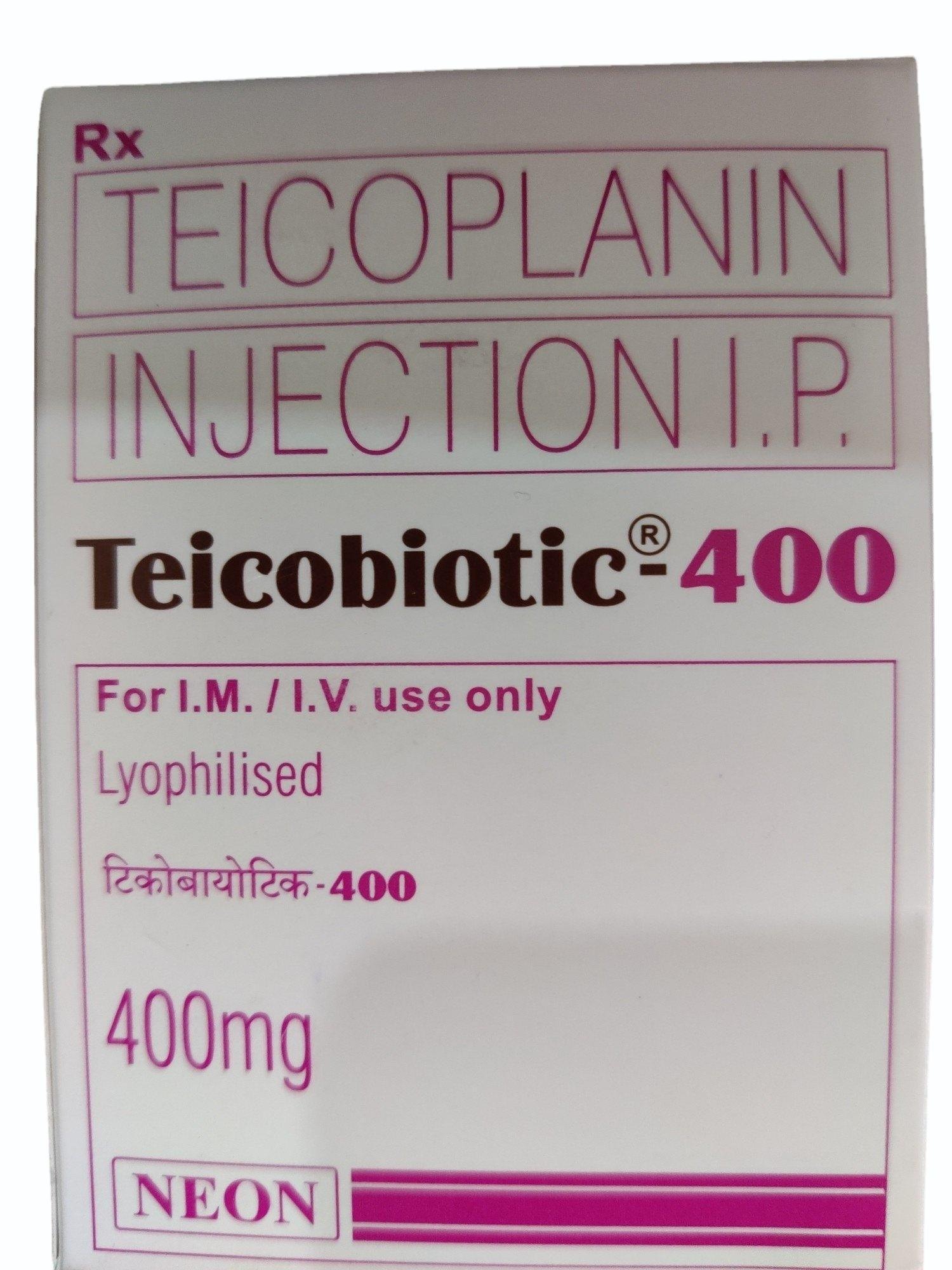
Teicobiotic Neon Teicoplanin 400 mg Injection
Price 2700.0 INR
Minimum Order Quantity : 100
Quantity : 1 Vial of 400 mg
Dosage Form : Injection, for IV use
Pacakaging (Quantity Per Box) : Each box contains 1 vial
Storage Instructions : Store below 25C, protect from light and moisture
Myo Pyrolate 5ml Glycopyrrolate + Neostiqmine Injection
Price 70.0 INR
Minimum Order Quantity : 100
Quantity : 5ml
Dosage Form : Injection
Pacakaging (Quantity Per Box) : 5 ampoules of 5ml each
Storage Instructions : Store in a cool, dry place, protected from light
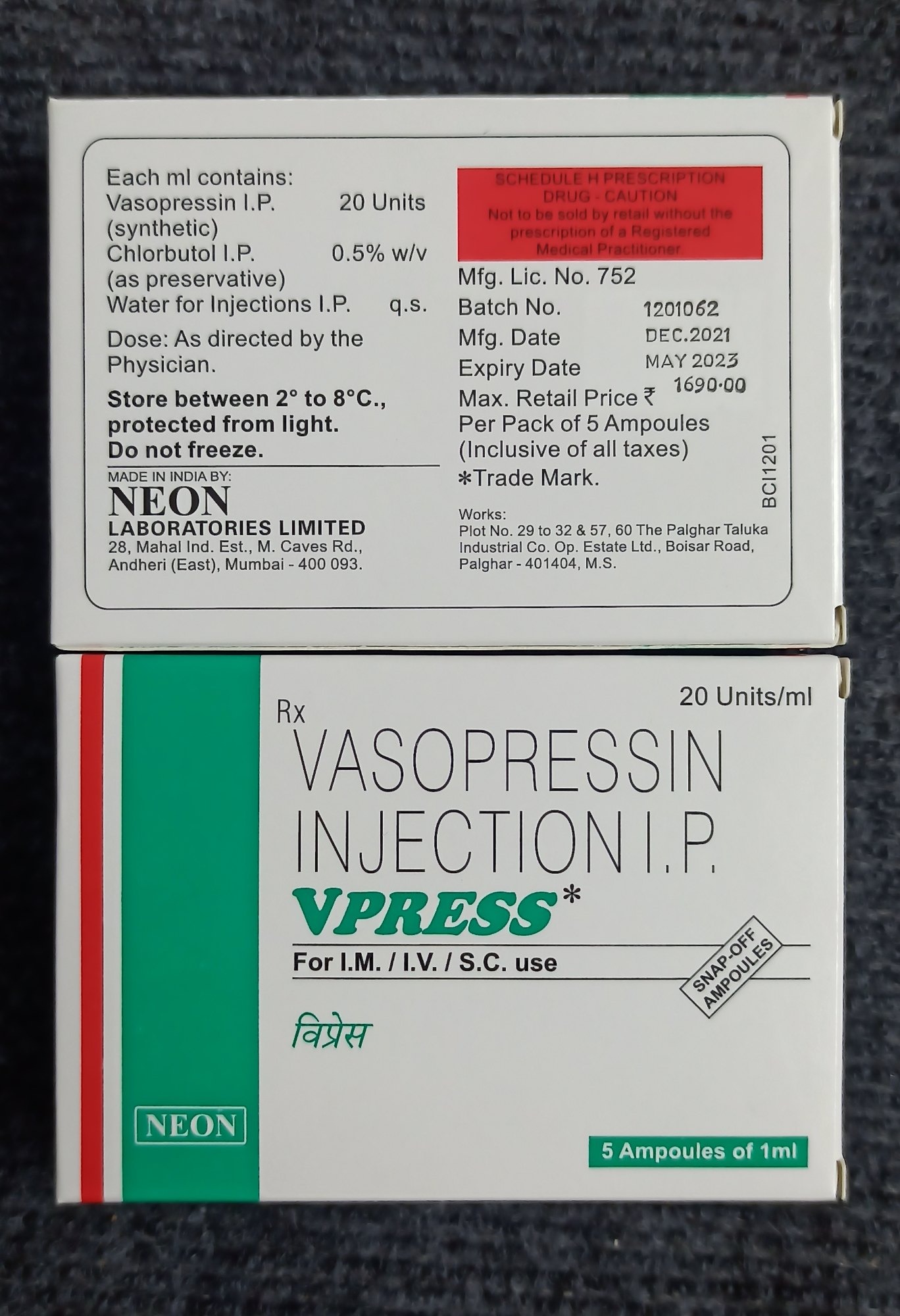
Vasopressin (20iu) Suspension Vpress Injection
Price 370.0 INR
Minimum Order Quantity : 100
Quantity : 1 vial
Dosage Form : Injection
Pacakaging (Quantity Per Box) : As per pack (usually 1 or 10 vials per box)
Storage Instructions : Store in a cool & dry place, protect from light
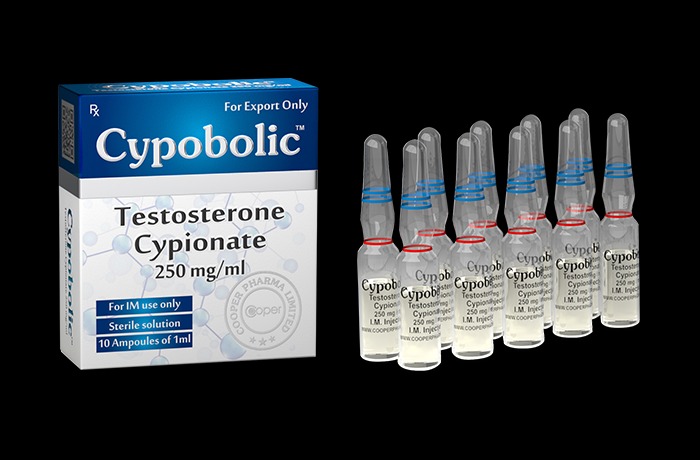
Testos-terone Cypionate 250 Mg Injection
Price 1200.0 INR
Minimum Order Quantity : 10
Quantity : 1 ampoule (1 ml)
Dosage Form : Injectable solution
Pacakaging (Quantity Per Box) : 1 ampoule per box
Storage Instructions : Store below 25C, protect from light and moisture

 Send Inquiry
Send Inquiry



 Send Inquiry
Send Inquiry Send SMS
Send SMS