Urofollitropin Injections Bp 75 Iu
Urofollitropin Injections Bp 75 Iu Specification
- Route of Administration
- Intramuscular/Subcutaneous
- Composition
- Urofollitropin
- Prescription/Non prescription
- Prescription
- Product Name
- Urofollitropin Injections Bp 75 Iu
- Drug Class
- Gonadotropins
- Warning
- For hospital use only
- Strength
- 75 IU
- Packaging Type
- Vial
- Form
- Injection
- Dosage
- As directed by physician
- Usage
- Female infertility, Assisted reproductive techniques
- Storage
- Store in a refrigerator (2°C - 8°C). Do not freeze.
- Shelf Life
- 24 months
Urofollitropin Injections Bp 75 Iu Trade Information
- Supply Ability
- 100 Per Day
- Delivery Time
- 7 Days
- Sample Available
- No
- Sample Policy
- Contact us for information regarding our sample policy
About Urofollitropin Injections Bp 75 Iu
| Dose |
|
| Packaging Size |
|
| Packaging Type |
|
| Drug Strength |
|
| Product Type |
|
| Salt |
|
| Minimum Order Quantity |
|
Product Specification
Product DescriptionOvitrop HP 75 Injection belongs to the class of drugs called hormones used in the treatment of female infertility in women whose ovaries do not produce eggs and in women receiving treatments like IVF (in-vitro fertilization). Infertility is unable to become pregnant after trying for a year. Female infertility may be caused due to different factors affecting ovulation (release of an egg from the ovary), fertilization (fusion of sperm and egg) and implantation (fertilized egg attaches to uterus lining).
| Brand | sun |
| Usage/Application | Clinical |
| Composition | Urofollitropin (75IU) |
| Injectable Form | Liquid |
| Dose | 75 iu |
| Medicine Type | Injection |
| Product Type | Finished Product |
| Country of Origin | Made in India |
Effective Solution for Female Infertility
Urofollitropin Injections BP 75 IU offers a reliable treatment option for women facing infertility, especially during assisted reproductive procedures. By mimicking natural hormones, it stimulates ovarian follicle development to improve the chances of conception. Each vial provides a precisely measured dose, supporting controlled and consistent therapy tailored to individual needs.
Safe and Controlled Hospital Administration
This injection is restricted to hospital use to ensure patient safety and optimal results. The medication must be administered by qualified medical professionals via intramuscular or subcutaneous routes, reducing the risk of improper dosing or complications. Required refrigeration helps maintain the efficacy and stability of the Urofollitropin.
Strict Storage Requirements for Maximum Potency
Storing Urofollitropin Injections BP 75 IU correctly is crucial for preserving their effectiveness. The vials must be kept refrigerated between 2C and 8C, away from freezing temperatures. Strict adherence to these guidelines ensures the medicine remains potent throughout its 24-month shelf life.
FAQ's of Urofollitropin Injections Bp 75 Iu:
Q: How should Urofollitropin Injections BP 75 IU be administered?
A: Urofollitropin Injections BP 75 IU should be administered either intramuscularly or subcutaneously, strictly under the supervision of a healthcare professional within a hospital setting.Q: What is the main benefit of using Urofollitropin Injections in infertility treatment?
A: The primary benefit is its ability to stimulate the ovaries, promoting the development of eggs, thereby increasing the chances of pregnancy for women undergoing assisted reproductive techniques.Q: When should Urofollitropin Injections be taken during infertility treatment?
A: The timing and dosage are determined by your physician, based on your specific reproductive cycle and treatment plan. Always follow the schedule provided by your healthcare provider.Q: Where should the product be stored to maintain its effectiveness?
A: Vials of Urofollitropin Injections must be stored in a refrigerator at 2C - 8C and must not be frozen, to preserve the potency and expiration of the medication.Q: What process is involved before starting Urofollitropin treatment?
A: Before initiating treatment, a thorough medical evaluation and hormonal assessment are conducted by your doctor, who will also explain the procedure and potential outcomes.Q: Is a prescription required for Urofollitropin Injections BP 75 IU?
A: Yes, this medication is only available by prescription and is intended strictly for use in hospitals by medical professionals.Q: What precautions should be taken during the usage of this injection?
A: It is crucial to follow your physician's instructions carefully, report any side effects, and ensure the medicine is always administered by qualified healthcare personnel in an appropriate clinical setting.

Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Pharmaceutical Tablets Category
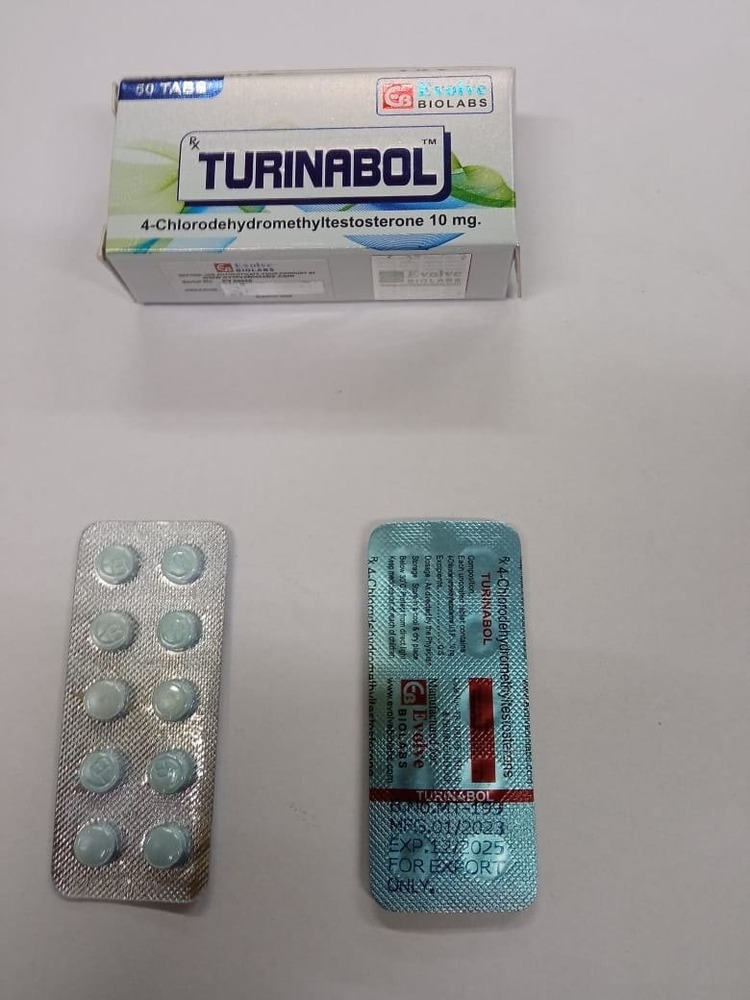
Turina-bol (Tbol) 4-Chlorodehydro-methyl) 10mg
Price 1000.0 INR / Strip
Minimum Order Quantity : 100 Strips
We Mainly Deal in International Market

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS